Safety
Demonstrated Safety Profile
The safety of ORGOVYX was evaluated in HERO, a randomized (2:1), open-label, clinical study (48 weeks) in patients with advanced prostate cancer1*
Serious adverse reactions occurred in 12% of patients receiving ORGOVYX1
- Serious adverse reactions in ≥0.5% of patients included: myocardial infarction (0.8%), acute kidney injury (0.6%), arrhythmia (0.6%), hemorrhage (0.6%), and urinary tract infection (0.5%)
Fatal adverse reactions occurred in 0.8% of patients receiving ORGOVYX. Fatal and non-fatal myocardial infarction and stroke were reported in 2.7% of patients receiving ORGOVYX1
Permanent discontinuation of ORGOVYX due to an adverse reaction occurred in 3.5% of patients1
Dosage interruptions of ORGOVYX due to an adverse reaction occurred in 2.7% of patients1
Adverse reactions (≥10%) of patients with advanced prostate
cancer who received ORGOVYX in HERO¹
- Clinically relevant adverse reactions in <10% of patients who received ORGOVYX included increased weight, insomnia, gynecomastia, hyperhidrosis, depression, decreased libido, and angioedema¹
Select laboratory abnormalities (≥15%) that worsened from baseline in patients with
advanced prostate cancer who received ORGOVYX in HERO¹
Exploratory safety analysis of unadjudicated major adverse
cardiovascular events1-3
A prespecified exploratory safety analysis determined the incidence of unadjudicated MACE using a MACE composite query including non-fatal myocardial infarction, non-fatal stroke, and death from any cause²
- 2.9% (n=18) of patients on ORGOVYX experienced a MACE, with 1.3% (n=8) of patients experiencing a grade 3/4 MACE
- 6.2% (n=19) of patients on leuprolide experienced a MACE, with 1.3% (n=4) of patients experiencing a grade 3/4 MACE
In an FDA analysis reported in the US Prescribing Information, fatal and non-fatal myocardial infarction and stroke were reported in 2.7% of patients receiving ORGOVYX¹
Patients with certain significant cardiovascular risk conditions were excluded from the HERO study. Some of the prior conditions excluded were³:
- Myocardial infarction or thromboembolic events within 6 months
- Arrhythmias
- Uncontrolled hypertension
Exploratory analysis of cumulative incidence of major adverse cardiovascular events through Week 48²
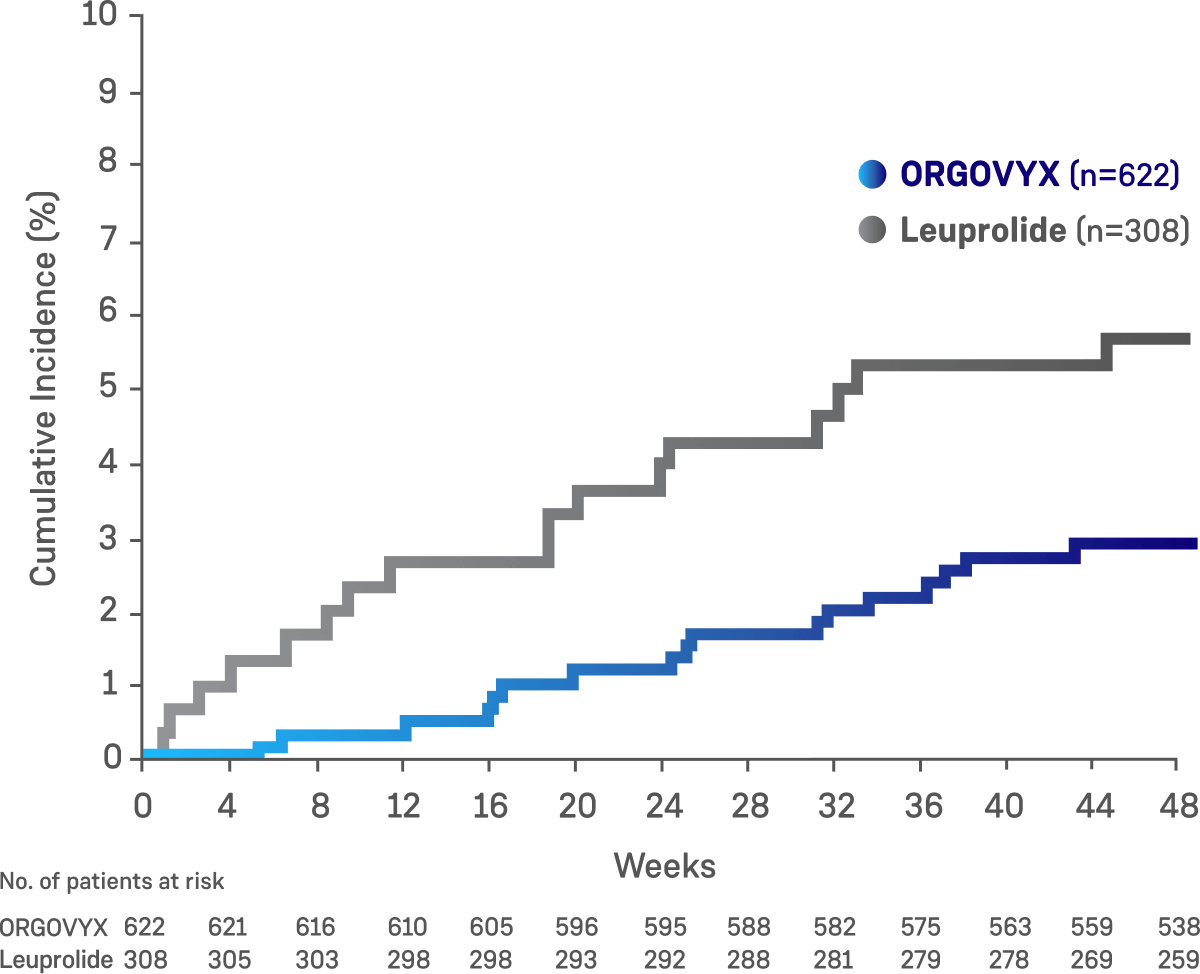
This exploratory prespecified safety analysis was not an alpha-protected endpoint in the study, and no adjustments were made for multiplicity of testing and therefore, no statistical conclusions can be drawn.
Limitations²
- The study did not include patients at high risk of cardiovascular events, which limits generalizability of the results to this population
- Study design did not include a prespecified endpoint with alpha control, adjudication of cardiac events, or a time-to-event analysis in the intent-to-treat population to compare the groups
- The absence of a placebo group limits the overall cardiac safety assessment of ORGOVYX
MACE=major adverse cardiovascular event.
Cardiovascular risk factors at baseline were well balanced between treatment arms of the HERO study²
Percentages of patients with these risk factors across the two treatment groups were²:
- 80% of patients had cardiovascular or cerebrovascular risk factors, such as diabetes or hypertension
- 67% of patients had lifestyle risk factors, such as a history of smoking or obesity
- 14% of patients had a history of myocardial infarction or stoke
NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for prostate cancer recommend screening and monitoring for cardiovascular disease, among other factors, when prescribing androgen deprivation therapy.4
NCCN=National Comprehensive Cancer Network.
Learn About Once-a-Day
Dosing With ORGOVYX
IMPORTANT SAFETY INFORMATION
Contraindication
ORGOVYX is contraindicated in patients with severe hypersensitivity to relugolix or to any of the product components.
Warnings and Precautions
QT/QTc Interval Prolongation: Androgen deprivation therapy, such as ORGOVYX may prolong the QT/QTc interval. Providers should consider whether the benefits of androgen deprivation therapy outweigh the potential risks in patients with congenital long QT syndrome, congestive heart failure, or frequent electrolyte abnormalities and in patients taking drugs known to prolong the QT interval. Electrolyte abnormalities should be corrected. Consider periodic monitoring of electrocardiograms and electrolytes.
Hypersensitivity: Angioedema was reported in 0.2% of patients treated with ORGOVYX in HERO. Hypersensitivity reactions, including pharyngeal edema and other serious cases of angioedema, have been reported post-marketing with ORGOVYX. Advise patients who experience any symptoms of hypersensitivity to temporarily discontinue ORGOVYX and promptly seek medical care. Discontinue ORGOVYX for severe hypersensitivity reactions and manage as clinically indicated.
Embryo-Fetal Toxicity: The safety and efficacy of ORGOVYX have not been established in females. Based on findings in animals and mechanism of action, ORGOVYX can cause fetal harm and loss of pregnancy when administered to a pregnant female. Advise males with female partners of reproductive potential to use effective contraception during treatment and for 2 weeks after the last dose of ORGOVYX.
Laboratory Testing: Therapy with ORGOVYX results in suppression of the pituitary gonadal system. Results of diagnostic tests of the pituitary gonadotropic and gonadal functions conducted during and after ORGOVYX may be affected. The therapeutic effect of ORGOVYX should be monitored by measuring serum concentrations of prostate-specific antigen (PSA) periodically. If PSA increases, serum concentrations of testosterone should be measured.
Adverse Reactions
Serious adverse reactions occurred in 12% of patients receiving ORGOVYX. Serious adverse reactions in ≥0.5% of patients included myocardial infarction (0.8%), acute kidney injury (0.6%), arrhythmia (0.6%), hemorrhage (0.6%), and urinary tract infection (0.5%). Fatal adverse reactions occurred in 0.8% of patients receiving ORGOVYX including metastatic lung cancer (0.3%), myocardial infarction (0.3%), and acute kidney injury (0.2%). Fatal and non-fatal myocardial infarction and stroke were reported in 2.7% of patients receiving ORGOVYX.
Most common adverse reactions (≥10%) and laboratory abnormalities (≥15%) in patients receiving ORGOVYX were hot flush (54%), glucose increased (44%), triglycerides increased (35%), musculoskeletal pain (30%), hemoglobin decreased (28%), alanine aminotransferase increased (27%), fatigue (26%), aspartate aminotransferase increased (18%), constipation (12%), and diarrhea (12%).
Drug Interactions
Co-administration of ORGOVYX with an oral P-gp inhibitor increases relugolix exposure, which may increase the risk of adverse reactions associated with ORGOVYX. Avoid co-administration of ORGOVYX with oral P-gp inhibitors. If co-administration is unavoidable, take ORGOVYX first and separate dosing by at least 6 hours. Monitor patients for increased adverse reactions. Treatment with ORGOVYX may be interrupted for up to 2 weeks if a short course of treatment with a P-gp inhibitor is required. Resume ORGOVYX after the P-gp inhibitor is discontinued. If treatment with ORGOVYX is interrupted for greater than 7 days, restart ORGOVYX with a 360 mg loading dose on the first day and continue with 120 mg once daily.
Co-administration of ORGOVYX with a combined P-gp and strong CYP3A inducer decreases relugolix exposure, which may reduce the effects of ORGOVYX. Avoid co-administration of ORGOVYX with combined P-gp and strong CYP3A inducers. If co-administration is unavoidable, increase the ORGOVYX dose to 240 mg once daily. After discontinuation of the combined P-gp and strong CYP3A inducer, resume the recommended ORGOVYX dose of 120 mg once daily.
Please see full Prescribing Information for ORGOVYX.
INDICATION
ORGOVYX® (relugolix) is a gonadotropin-releasing hormone (GnRH) receptor antagonist indicated for the treatment of adult patients with advanced prostate cancer.
References: 1. ORGOVYX (relugolix). Prescribing information. Sumitomo Pharma America, Inc.; 2025. 2. Shore ND, Saad F, Cookson MS, et al. Oral relugolix for androgen-deprivation therapy in advanced prostate cancer. N Engl J Med. 2020;382(23):2187-2196. doi:10.1056/NEJMoa2004325 3. Shore ND, Saad F, Cookson MS, et al. Protocol for: Oral relugolix for androgen-deprivation therapy in advanced prostate cancer. N Engl J Med. 2020;382(23)(suppl):2187-2196. doi:10.1056/NEJMoa2004325 4. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Prostate Cancer V.5.2026. © National Comprehensive Cancer Network, Inc. 2026. All rights reserved. Accessed January 23, 2026. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.


